
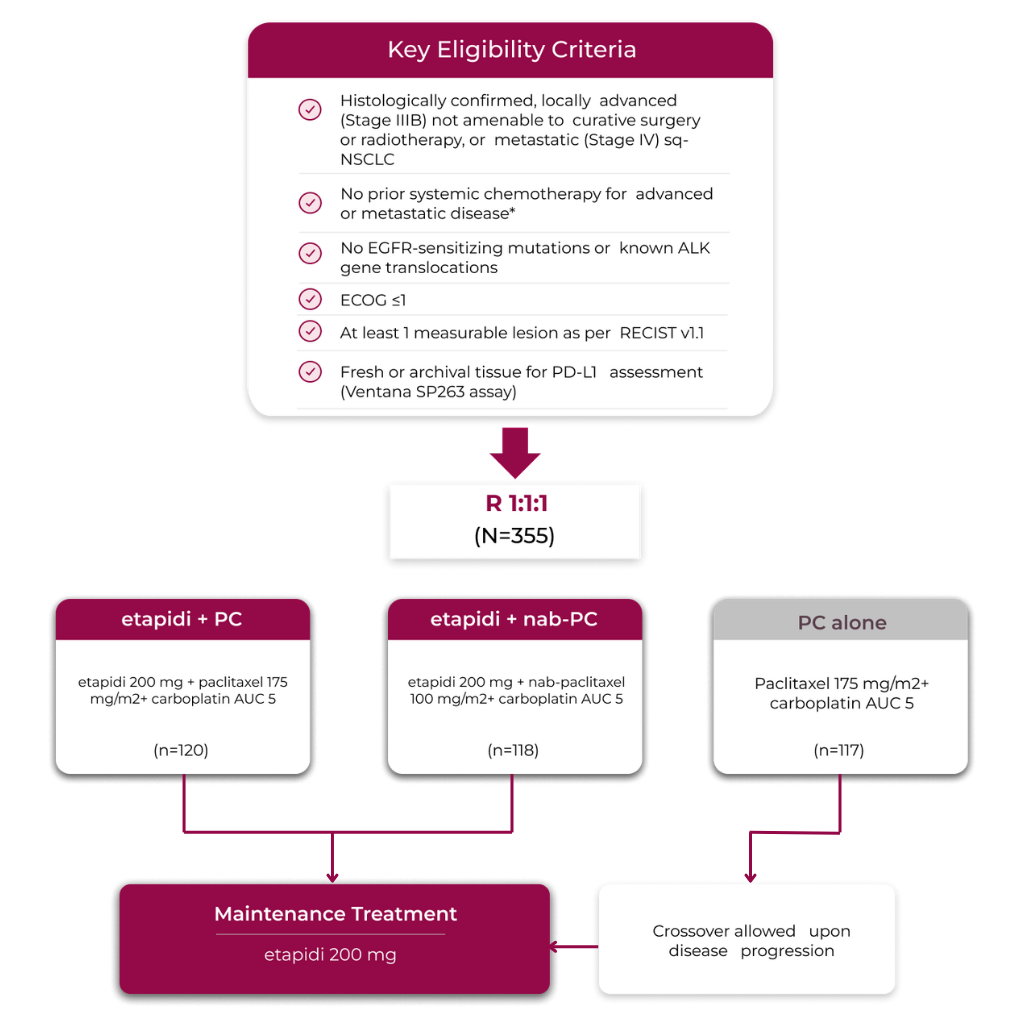
RATIONALE 307: A randomized phase 3 trial in nearly 360 patients with 1L, locally advanced or metastatic Non-squamous NSCLC
This global trial was a randomized, double-blind, placebo-controlled, phase 3 trial conducted in 16 countries at 162 sites across multiple continents: Asia, Europe, North America, and Oceania*
In the chemo arm, 77 (63.6%) patients received subsequent anti-PD-(L)1 therapy, including 71 (58.7%) who crossed over to tislelizumab in-study; analyses adjusted for in-study crossover effect with the two-stage method further confirmed the OS benefits with tislelizumab + chemo (cross-over adjusted median OS of chemo: 16.0 mo; HRs 0.53 and 0.65, respectively
RATIONALE-307 was stratified by:
- Stage (IIIB vs IV)
- PD-L1 TC (<1% vs 1%-49% vs ≥50%)
etapidi + chemotherapy was tested across a broad range of patients with a PD-L1 score ≥1%4
| Baseline characteristics for patients with advanced Squamous NSCLC | etapidi + PP (n=223) | etapidi + nab-PC (n=111) | PC (n=121) | |
|---|---|---|---|---|
| Median age, years (range) | 60 (41–74) | 63 (38–74) | 62 (34–74) | |
| Sex, n (%) | Male | 107 (89.2) | 112 (94.1) | 111 (91.7) |
| Tobacco use, n (%) | Current/former | 96 (80.0) | 107 (89.9) | 98 (81.0) |
| Never | 24 (20.0) | 12 (10.1) | 23 (19.0) | |
| ECOG PS, n (%) | 0 | 31 (25.8) | 22 (18.5) | 32 (26.4) |
| 1 | 89 (74.2) | 97 (81.5) | 89 (73.6) | |
| Disease stage, n (%) | IIIB | 38 (31.7) | 40 (33.6) | 44 (36.4) |
| IV | 82 (68.3) | 79 (66.4) | 77 (63.6) | |
| Location of distant metastases, n (%) | Bone | 24 (20.0) | 16 (13.4) | 21 (17.4) |
| Liver | 15 (12.5) | 15 (12.6) | 14 (11.6) | |
| Brain | 2 (1.7) | 3 (2.5) | 1 (0.8) | |
| PD-L1 on TC, n (%) | <1% | 48 (40.0) | 47 (39.5) | 49 (40.5) |
| ≥1% | 72 (60.0) | 72 (60.5) | 72 (59.5) | |
| 1–49% | 30 (25.0) | 30 (25.2) | 31 (25.6) | |
| ≥50% | 42 (35.0) | 42 (35.3) | 41 (33.9) | |
References: 1. etapidi. Prescribing Information. BeOne Medicines USA, Inc.; 2025. 2. Zhang T, et al. Cancer Immunol Immunother. 2018;67:1079-1090. 3. Hong Y, et al. FEBS Open Bio. 2021;11:782-792. 4. Wang J, et al. JAMA Oncol. 2021 May 1;7(5):709-717. 5. Lu S, et al. J Thorac Oncol. 2021 Sep;16(9):1512-1522. 6. Xu J, et al. Lancet Oncol. 2023; 24: 483–95. 7. Qiu M, et al. BMJ. 2024 May 28:385:e078876. 9. Cheng Y, et al. J Thorac Oncol. 2024 Jul;19(7):1073-1085.
