
RATIONALE 304: A randomized phase 3 trial in nearly 334 patients with 1L, locally advanced or metastatic Non-squamous NSCLC
RATIONALE-304 was a randomised, open-label, multi-centre, Phase 3 study investigating the efficacy and safety of ETAPIDI plus platinum-based chemotherapy vs platinum-based chemotherapy alone as a first-line treatment for patients with advanced non-squamous NSCLC.
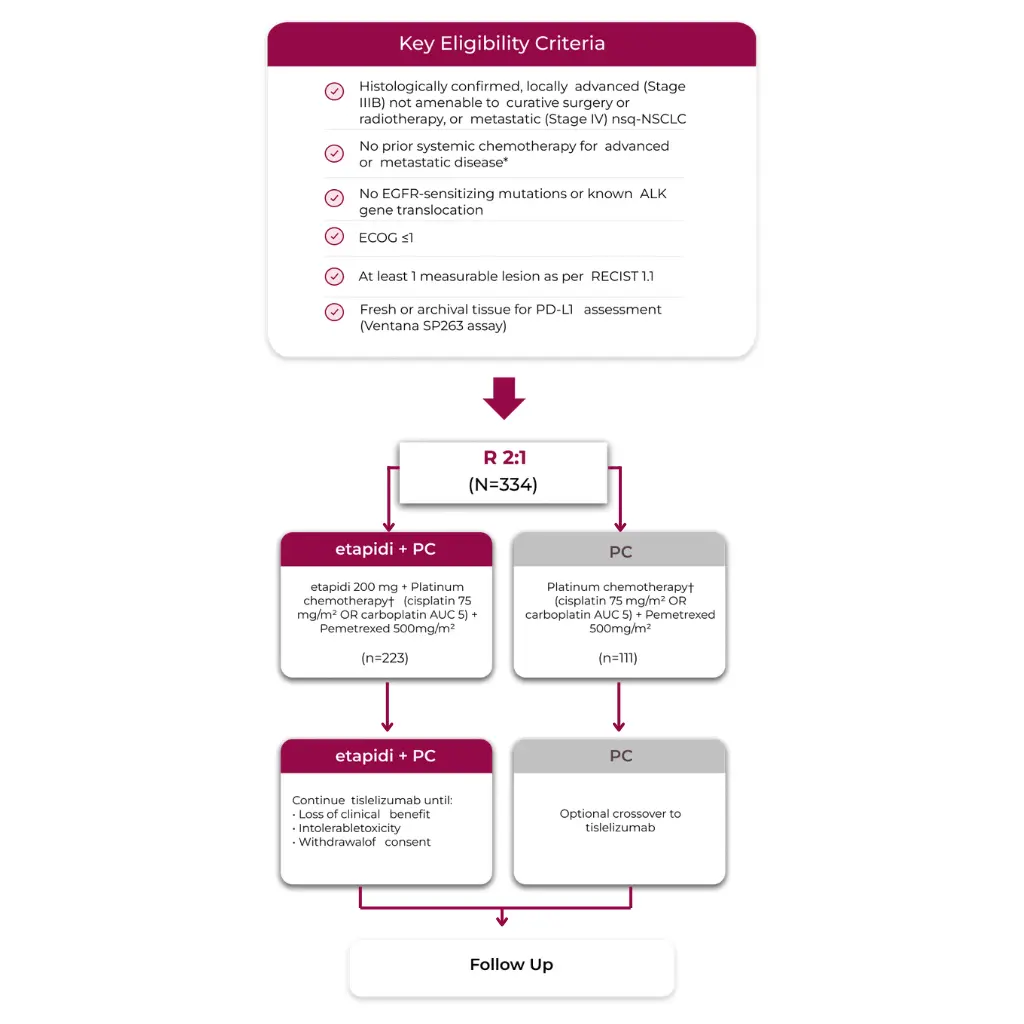
Stratification factors
- Disease stage (IIIB vs IV)
- PD-L1 TC expression (<1% vs 1%-49% vs ≥50%)
Efficacy endpoints
- Primary endpoint: IRC-assessed PFS
- Secondary endpoints: ORR, DoR, OS, safety
profile, and HRQoL
RATIONALE-304 featured a broad range of patients, including those with difficult-to-treat characteristics
| Baseline characteristics for patients with advanced Non-squamous NSCLC |
etapidi + PP (n=223) | Chemotherapy Alone (n=111) |
|---|---|---|
| Median age, y (range) | 60 (27-75) | 61 (25-74) |
| Age group, n (%) | ||
| <65 y | 163 (73.1) | 74 (66.7) |
| ≥65 y | 60 (26.9) | 37 (33.3) |
| Sex, n (%) | ||
| Male | 168 (75.3) | 79 (71.2) |
| Female | 55 (24.7) | 32 (28.8) |
| Smoking status, n (%) | ||
| Former | 115 (51.6) | 53 (47.7) |
| Current | 32 (14.3) | 13 (11.7) |
| Never | 76 (34.1) | 45 (40.5) |
| ECOG performance status, n (%) | ||
| 0 | 54 (24.2) | 24 (21.6) |
| 1 | 169 (75.8) | 87 (78.4) |
| Disease stage at baseline, n (%) | ||
| Stage IIIB | 40 (17.9) | 21 (18.9) |
| Stage IV | 183 (82.1) | 90 (81.1) |
| Histologic type, n (%) | ||
| Adenocarcinoma | 215 (96.4) | 107 (96.4) |
| Mixed adenosquamous | 1 (0.4) | 2 (1.8) |
| Other | 7 (3.1) | 2 (1.8) |
References: 1. TEVIMBRA. Prescribing Information. BeOne Medicines USA, Inc.; 2025. 2. Shen L, Kato K, Kim S-B, et al; RATIONALE-302 Investigators. J Clin Oncol. 2022;40(26):3065-3076. doi:10.1200/JCO.21.01926
