
No new safety signals identified in 1L GC/GEJC *1
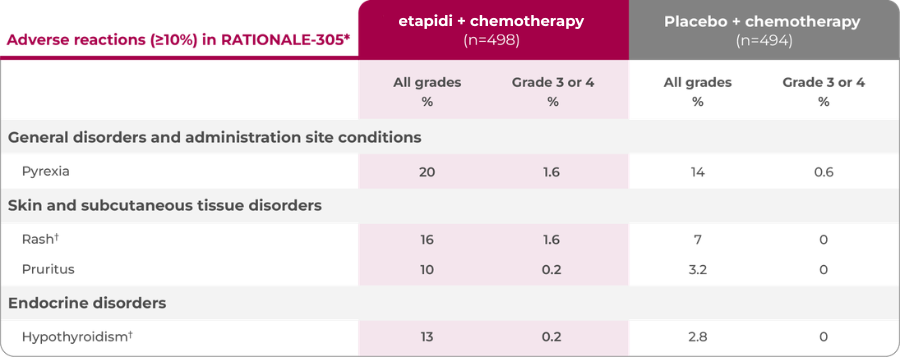
Adverse reactions (≥10%) in RATIONALE-305†
Discontinuation:
- Permanent discontinuation of etapidi in the etapidi + chemotherapy arm due to an adverse drug reaction occurred in 16% of patients
- Adverse drug reactions that resulted in permanent discontinuation in ≥1% of patients were death, fatigue, and pneumonitis
imAEs leading to discontinuation2:
- 3.1% of patients receiving etapidi
- imAEs leading to discontinuation of etapidi included immune-mediated pneumonitis (5 patients [2%]), immune-mediated myositis/rhabdomyolysis (2 patients [0.8%]), and immune-mediated myocarditis (1 patient [0.4%])
*With a difference between arms of ≥5% for all grades or ≥2% for grades 3 and 4.
†Represents a composite of multiple, related preferred terms.
1L, first line; 2L, second line; ESCC, esophageal squamous cell carcinoma; imAE, immune-mediated adverse event; PD-1, programmed death receptor 1; PD-L1, programmed death ligand 1; TEAE, treatment-emergent adverse event.
References: 1. etapidi. Prescribing Information. BeOne Medicines USA, Inc.; 2025. 2. Data on file. BeOne, Ltd.
