
RATIONALE-305: A global, randomized, double-blind, placebo-controlled, phase 3 trial in nearly 1,000 patients with 1L, unresectable or metastatic, HER2-negative GC or GEJC1-3
This global trial was a randomized, double-blind, placebo-controlled, phase 3 trial conducted in 16 countries at 162 sites across multiple continents: Asia, Europe, North America, and Oceania*
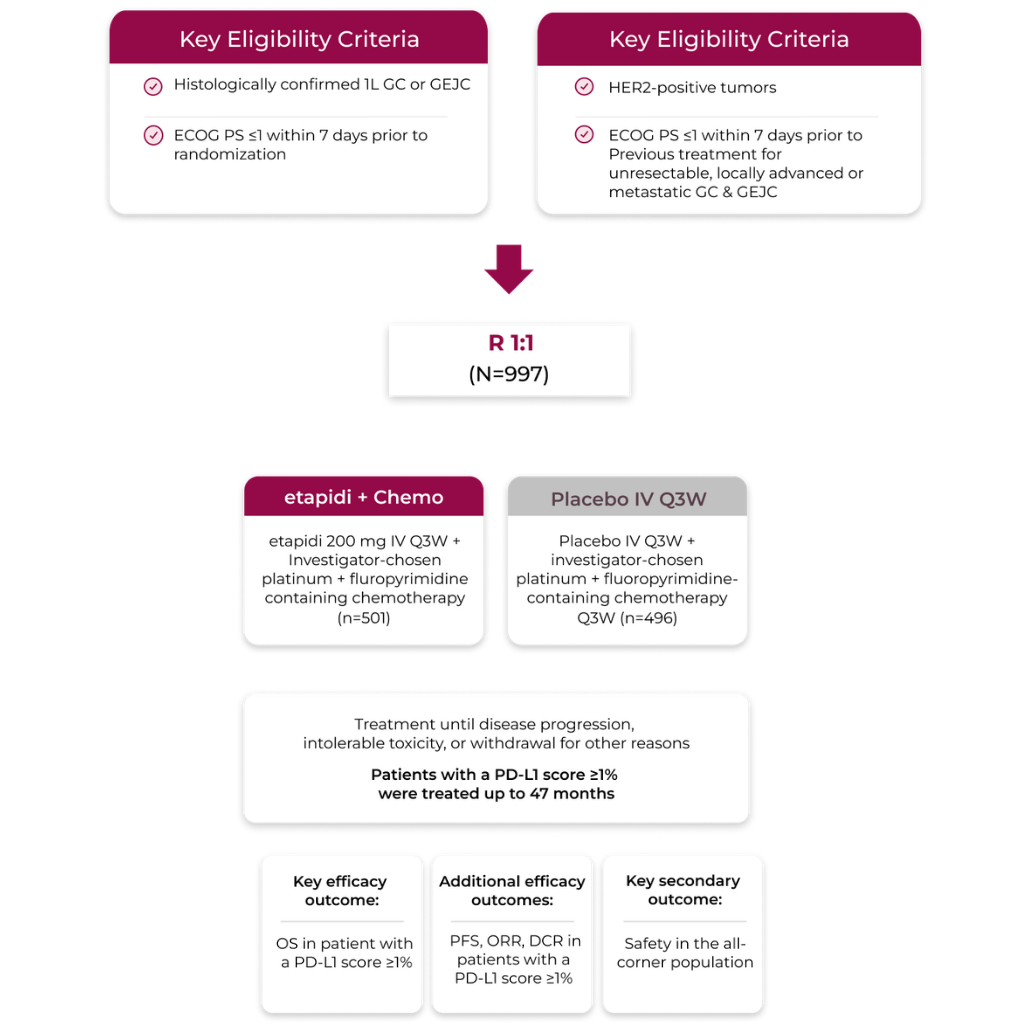
- Key efficacy analysis in patients with a PD-L1 score ≥1%*: n=885
- Safety analysis population: n=992
RATIONALE-305 was designed to show consistent benefit across a broad range of patient types. The study was stratified by:
- Region of enrollment
- Peritoneal metastases (yes/no)
- Investigator-chosen chemotherapy
- PD-L1 score (PD-L1 ≥5% vs <5%)
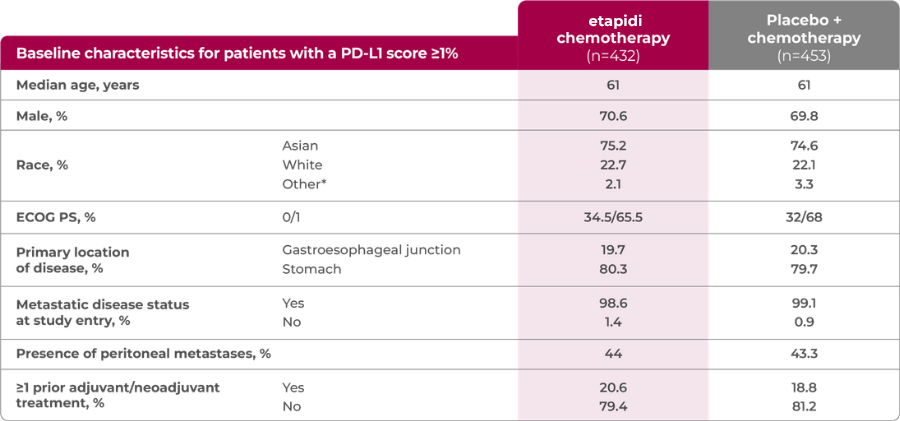
RATIONALE-305 featured a broad range of patients, including those with difficult-to-treat characteristics3
You may use your preferred PD-L1 testing method to identify patients in clinical practice
*The key efficacy analysis was a retrospective subgroup analysis of RATIONALE-305 in patients with a PD-L1 score ≥1%. PD-L1–negative patients were excluded from this analysis population due to TEVIMBRA + chemotherapy being indicated in patients whose tumors expressed PD-L1 (≥1).
†The race subcategory “Other” included not reported, unknown, and other.
1L, first line; CPS, combined positive score; DCR, disease control rate; ECOG, Eastern Cooperative Oncology Group; GC, gastric cancer; GEJC, gastroesophageal junction cancer; HER2, human epidermal growth growth factor 2; IV, intravenous; ORR, overall response rate; OS, overall survival; PD-L1, programmed death ligand 1; PFS, progression-free survival; PS, performance status; Q3W, every 3 weeks; R, randomization; TAP, tumor area positivity.
References: 1. etapidi. Prescribing Information. BeOne Medicines USA, Inc.; 2025. 2. Qiu MZ, Oh DY, Kato K, et al; RATIONALE-305 Investigators. BMJ. 2024;385:e078876. doi:10.1136/bmj-2023-078876 3. Moehler M, Oh DY, Kato K, et al. Adv Ther. 2025;42(5):2248-2268. doi:10.1007/s12325-025-03133-7 4. Raymond E, Xu J, Kato K, et al. Abstract presented at: ESMO Gastrointestinal Cancers Congress, June 26-29, 2024; Munich, Germany. Abstract 395MO.
